Pressure
You’ve heard of pressure many times throughout AP Chem, but what actually is it?
Pressure is a measure of force applied over a certain surface area.

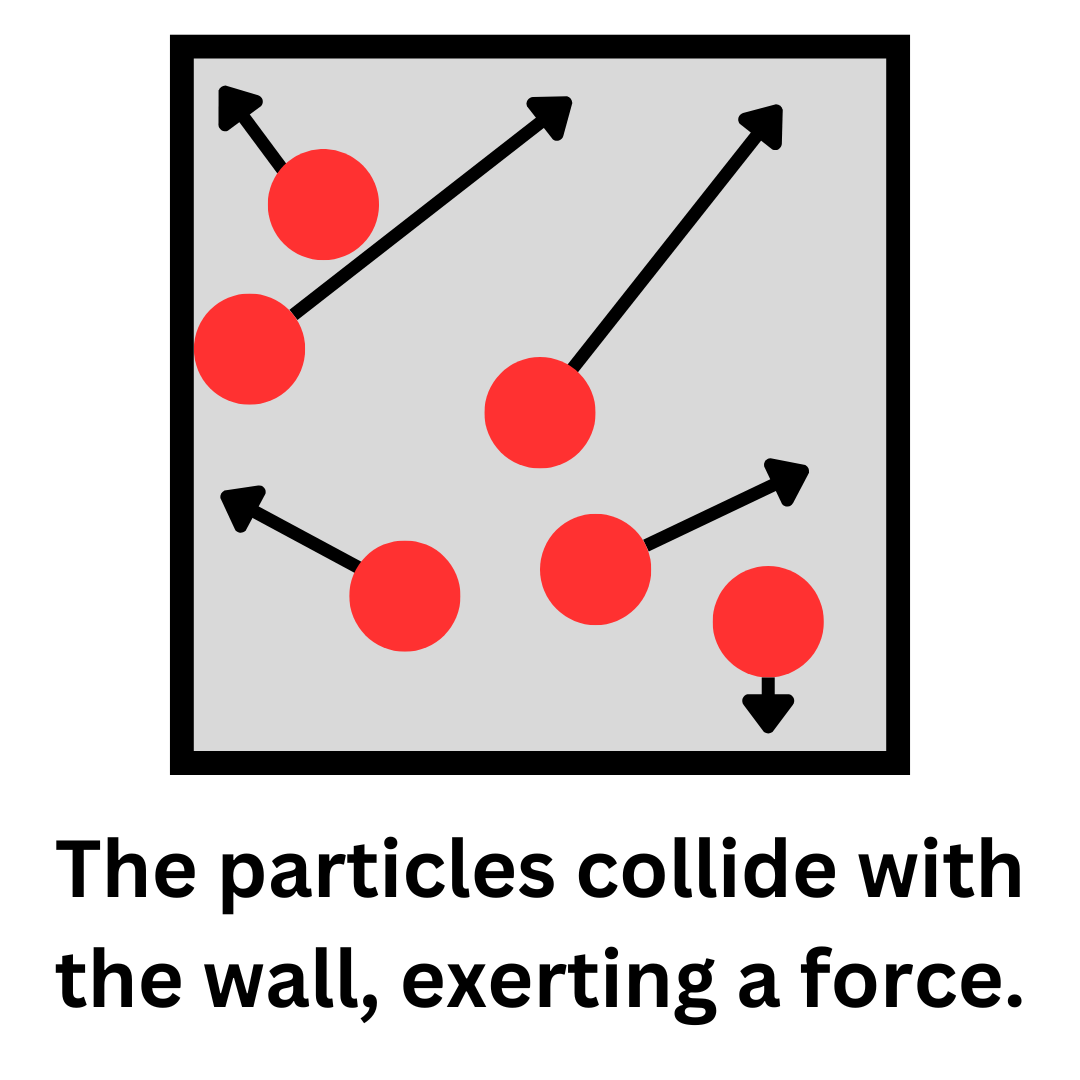
At a molecular level, gas particles collide with the walls of their container. During these collisions, the gas molecules exert a force on the wall. Pressure is not a measure of individual collisions with the wall. Instead, it measures the total force applied by all of the molecules over the entire surface area of the container.
Measuring Pressure
We commonly use atm as a unit of measuring pressure. 1 atm is the atmospheric pressure at sea level. 2 atm would be twice the atmospheric pressure at sea level.
Another common unit for pressure is mmhg, which stands for “millimeter of mercury (Hg)”. mmHg is also known as torr.
1 atm = 760 torr
The unit mmHg came from barometers, which are a common tool used to measure pressure. Here’s how they work.

As the atmospheric pressure increases, the mercury inside of the tube is pushed higher. It is somewhat similar to a thermometer.
Mercury is often the substance used in barometers. The pressure was measured by looking at the height of the mercury, thus mmHG = millimeter of mercury.

