Solid
– Definite (fixed) shape and volume
– Cannot be compressed
– Organized structure
– Least energy (compared to liquid and gas)
– Molecules are close together
– Molecules are locked in place
Liquid
– Definite volume
– No fixed shape (takes shape of container)
– Cannot be compressed
– More energy than solid, but less than gas
– Molecules are close together
– Molecules are free to slide past each other
Gas
– No fixed shape (takes shape of container)
– No fixed volume (takes volume of container)
– Compressible
– Most energy
– Molecules are evenly spread out
– Molecules are free to move in any direction
Phase Changes
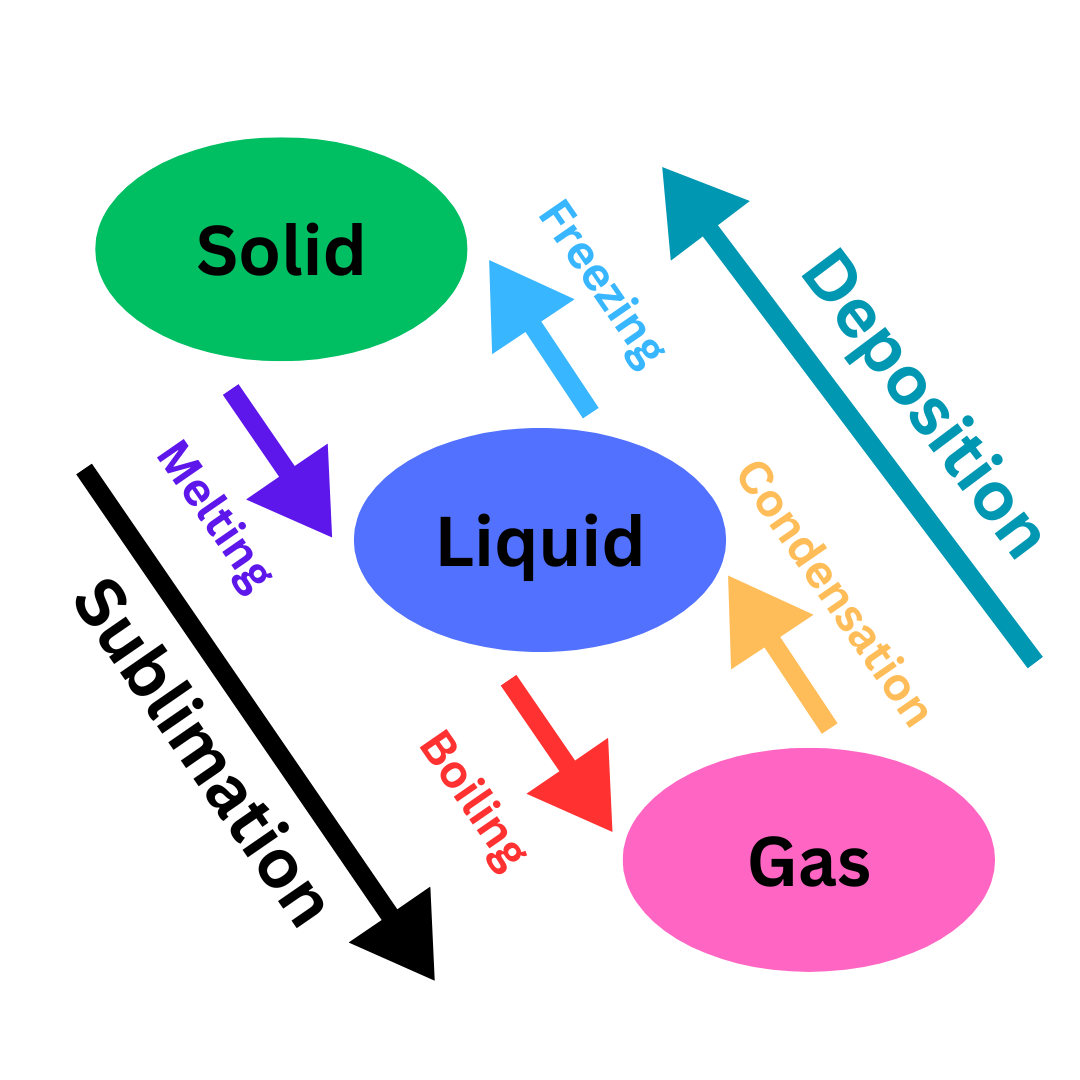
Melting: Heating a solid until it becomes a liquid.
Freezing: Cooling a liquid until it becomes a solid.
Boiling: Heating a liquid until it becomes a gas.
Condensation: Cooling a gas until it becomes a liquid.
Sublimation: Heating a solid so quickly that it goes directly to the gaseous state.
Deposition: Going directly from the gaseous to solid phase.
